
We use cookies to ensure our website works properly and to personalise your experience. Cookies policy

We use cookies to ensure our website works properly and to personalise your experience. Cookies policy
1Department of Pharmaceutics, Institute of Pharmacy,Shri Harishchandra Post Graduate College., Varanasi
2Department of Pharmaceutics, Institute of Pharmacy,Shri Harishchandra Post Graduate College., Varanasi
The present study was aimed at the formulation and evaluation of a polyherbal face pack for anti-acne and skin brightening effects using natural herbal ingredients. Various herbal ingredients such as multani mitti, neem, orange peel, aloe vera, tulsi, sandalwood, licorice, green tea, activated charcoal, and manjistha were selected based on their antimicrobial, anti-inflammatory, antioxidant, cleansing, and skin brightening properties. Three formulations (F1, F2, and F3) were prepared by dry mixing method using different concentrations of herbal ingredients. The prepared formulations were evaluated for various physicochemical and performance parameters including organoleptic properties, pH, spreadability, washability, drying time, skin irritation, flow properties, and stability studies. Preformulation studies confirmed acceptable particle size, good flowability, suitable moisture content, and compatibility of all herbal powders. Among all formulations, F2 exhibited better spreadability, ideal pH, good consistency, easy washability, and excellent stability without causing skin irritation. Stability studies showed no significant changes in color, odor, texture, or pH during storage. The synergistic action of herbal ingredients provided effective cleansing, oil control, anti-acne activity, moisturizing effect, antioxidant protection, and improvement in skin brightness. The study concluded that the developed polyherbal face pack is safe, stable, effective, and suitable for cosmetic skin care application as a natural alternative to synthetic cosmetic products.
The skin is the largest organ of the human body and serves as a protective barrier against environmental pollutants, microorganisms, ultraviolet radiation, and physical damage. It also performs several important physiological functions such as thermoregulation, sensory perception, prevention of water loss, immune defense, and vitamin D synthesis. Proper maintenance of skin health is therefore essential for both physiological balance and cosmetic appearance.
Continuous exposure of facial skin to dust, sweat, excess sebum, pollutants, and cosmetic residues may lead to various skin problems such as acne, dullness, blackheads, whiteheads, rough texture, and uneven skin tone. Accumulation of dead skin cells on the skin surface interferes with normal skin renewal and reduces skin brightness. Therefore, regular cleansing and exfoliation are important for maintaining healthy and radiant skin.
Exfoliation is the process of removing dead skin cells from the outermost layer of the skin. It improves skin texture, unclogs pores, enhances skin smoothness, and promotes regeneration of new skin cells. Face scrubs are commonly used exfoliating cosmetic preparations that provide both cleansing and mechanical exfoliation through gentle rubbing action. However, many synthetic face scrubs available in the market contain harsh chemicals and artificial exfoliating particles that may cause skin irritation, dryness, and environmental pollution.
Due to the limitations associated with synthetic cosmetic products, there has been increasing interest in herbal cosmetic formulations prepared using natural ingredients. Herbal cosmetics are considered safer, biodegradable, eco-friendly, and more compatible with the skin. Natural ingredients such as papaya peel powder, guava leaf powder, rice flour, turmeric, and multani mitti possess antimicrobial, antioxidant, anti-inflammatory, cleansing, and exfoliating properties beneficial for skin care. Papaya peel contains papain enzyme which provides enzymatic exfoliation, while rice flour acts as a mild abrasive agent for removal of dead skin cells. Guava leaf powder and turmeric exhibit antibacterial and anti-inflammatory activities that help in maintaining healthy skin and reducing acne.
Therefore, the present study was undertaken to formulate and evaluate a herbal face scrub using natural exfoliating agents. The developed formulation was evaluated for various physicochemical and performance parameters such as pH, viscosity, spreadability, grittiness, washability, skin irritation, and stability in order to ensure safety, effectiveness, and cosmetic acceptability.
2. MATERIALS AND METHOD
2.1 MATERIALS
The herbal ingredients used for the preparation of the polyherbal face pack were procured from a local herbal supplier and were of analytical grade. All ingredients were properly dried and powdered before use.
Table: 1 list of materials
|
Sr. No |
Material / Ingredient |
Biological Source |
Use in Formulation |
|
1 |
Multani Mitti |
Natural clay (Fuller’s earth) |
Oil absorbing and cleansing agent |
|
2 |
Neem Powder |
Azadirachta indica leaves |
Antibacterial and anti-acne agent |
|
3 |
Orange Peel Powder |
Citrus sinensis peel |
Skin brightening and exfoliating agent |
|
4 |
Linseed Powder |
Linum usitatissimum seeds |
Moisturizing and skin nourishing agent |
|
5 |
Activated Charcoal |
Coconut shell derived carbon |
Detoxifying and impurity removing agent |
|
6 |
Licorice Powder |
Glycyrrhiza glabra roots |
Skin brightening and anti-pigmentation agent |
|
7 |
Green Tea Powder |
Camellia sinensis leaves |
Antioxidant and anti-inflammatory agent |
|
8 |
Aloe Vera Powder |
Aloe barbadensis leaf gel |
Moisturizing and soothing agent |
|
9 |
Tulsi Powder |
Ocimum sanctum leaves |
Antibacterial and anti-acne agent |
|
10 |
Manjistha Powder |
Rubia cordifolia roots |
Skin purification and complexion enhancer |
|
11 |
Sandalwood Powder |
Santalum album wood |
Cooling and skin brightening agent |
2.2 EQUIPMENT
Various laboratory instruments and equipment were used for the preparation and evaluation of the polyherbal face pack formulation.
Table : 2 list of equpments
|
Sr. No. |
Instrument / Equipment |
Company / Brand |
Model No. |
Purpose |
|
1 |
Digital Weighing Balance |
Shimadzu |
AY220 |
Accurate weighing of ingredients |
|
2 |
Mechanical Sieve Shaker |
Electrolab |
EMS-8 |
Particle size analysis |
|
3 |
Mortar and Pestle |
Borosil |
Porcelain Type |
Mixing of powders |
|
4 |
pH Meter |
Eutech Instruments |
pH 700 |
pH determination |
|
5 |
Magnetic Stirrer |
Remi |
2MLH |
Uniform mixing |
|
6 |
Hot Air Oven |
Thermolab |
THO-100 |
Moisture content determination |
|
7 |
Desiccator |
Borosil |
Glass Type |
Storage & moisture protection |
|
8 |
Glass Beakers |
Borosil |
Standard |
Sample preparation |
|
9 |
Measuring Cylinder |
Borosil |
100 ml |
Volume measurement |
|
10 |
Spatula |
Stainless Steel |
— |
Handling of powders |
2.3 PRE-FORMULATION STUDIES
Pre-formulation studies were carried out to evaluate the physicochemical properties of selected herbal powders before formulation development.
2.3.1 Organoleptic Evaluation
Procedure:
Step 1: About 500 mg of each herbal powder was taken on a watch glass.
Step 2: Color, odor, texture, and appearance were observed visually and by touch.
Parameers Observed:
•Color
•Odor
•Texture
•Appearance
2.3.2 Particle Size Determination (Sieve Analysis)
Procedure:
Step 1: 10 g of powder was weighed accurately.
Step 2: The powder was passed through sieve set containing 20, 40, and 60 mesh sieves.
Step 3: Sieving was carried out for 10 minutes using sieve shaker.
Step 4: Powder retained on each sieve was collected and weighed.
Purpose:
To obtain uniform particle size for smooth application.
2.3.3 Bulk Density
Procedure:
Step 1: 5 g powder was transferred into measuring cylinder.
Step 2: Bulk volume occupied by powder was noted.
Formula:
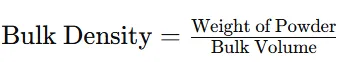
2.3.4 Tapped Density
Procedure:
Step 1: The measuring cylinder containing powder was tapped 100 times.
Step 2: Final tapped volume was recorded.
Formula:
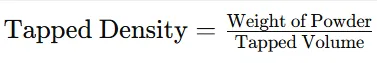
2.3.5 Angle of Repose
Procedure:
Step 1: 10 g powder was allowed to flow through a funnel.
Step 2: Height and radius of powder heap were measured.
Formula:
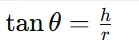
2.3.6 Carr’s Index
Definition:
Carr’s index indicates compressibility and flow property of powder.
Formula:

2.3.7 Hausner Ratio
Definition:
Hausner ratio is used to determine flowability of powder.
Formula:
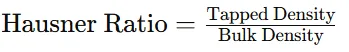
2.3.8 Moisture Content Determination
Procedure:
Step 1: 2 g powder was weighed and placed in petri dish.
Step 2: The sample was dried at 105°C for 30 minutes.
Step 3: Final weight was noted after drying.
Purpose:
To determine moisture content and stability of powder.
2.3.9 pH Determination
Procedure:
Step 1: 1 g powder was mixed with 10 mL distilled water.
Step 2: The mixture was stirred properly.
Step 3: pH was measured using digital pH meter.
Ideal Range:
Skin compatible pH range: 5–7
2.3.10 Compatibility Study
Procedure:
Step 1: Equal quantities of herbal powders were mixed.
Step 2: The mixture was stored for 24 hours at room temperature.
Step 3: The mixture was observed for color change, odor change, and lump formation.
Purpose:
To check compatibility between ingredients.
2.3.11 Phytochemical Analysis
Procedure:
Step 1: Herbal powder extracts were prepared using water or ethanol.
Step 2: The extracts were filtered.
Step 3: Different phytochemical tests were performed for alkaloids, flavonoids, tannins, phenols, saponins, and glycosides.
Step 4: Observations were recorded based on color change or precipitate formation.
2.4 FORMULATION OF POLYHERBAL FACE PACK
2.4.1 Formulation Table
Table No. 3: Composition of Polyherbal Face Pack Batches
|
Sr. No. |
Ingredient |
F1 (g) |
F2 (g) |
F3 (g) |
Role |
|
1 |
Multani Mitti |
25 g |
20 g |
15 g |
Oil absorbing agent |
|
2 |
Neem Powder |
10 g |
10 g |
10 g |
Anti-acne agent |
|
3 |
Orange Peel Powder |
10 g |
10 g |
10 g |
Brightening agent |
|
4 |
Linseed Powder |
5 g |
8 g |
12 g |
Moisturizing agent |
|
5 |
Activated Charcoal |
7 g |
5 g |
3 g |
Detoxifying agent |
|
6 |
Licorice Powder |
8 g |
8 g |
8 g |
Anti-pigmentation |
|
7 |
Green Tea Powder |
5 g |
7 g |
7 g |
Antioxidant |
|
8 |
Aloe Vera Powder |
10 g |
10 g |
10 g |
Soothing agent |
|
9 |
Tulsi Powder |
5 g |
7 g |
7 g |
Antibacterial |
|
10 |
Manjistha Powder |
5 g |
5 g |
8 g |
Skin purifier |
|
11 |
Sandalwood Powder |
10 g |
10 g |
10 g |
Cooling agent |
2.4.2 METHOD OF PREPARATION
The polyherbal face pack formulations (F1, F2, and F3) were prepared by dry mixing method using accurately weighed quantities of herbal powders.
Procedure:
Step 1: Weighing of Ingredients
All herbal ingredients were accurately weighed separately according to formulation composition using digital weighing balance.
Step 2: Sieving of Ingredients
All ingredients were passed through sieve no. 60 to obtain uniform particle size and remove lumps or impurities.
Step 3: Collection of Sieved Powders
The sieved powders were collected separately in clean and dry containers.
Step 4: Mixing by Geometric Dilution Method
The powders were mixed using geometric dilution method by gradual addition of ingredients with continuous mixing in mortar and pestle.
Step 5: Blending
The complete mixture was blended for 15–20 minutes to obtain uniform and homogeneous powder mixture.
Step 6: Evaluation of Physical Appearance
The prepared formulations were visually observed for color, texture, homogeneity, and absence of lumps.
Step 7: Packaging
The formulations were transferred into clean, dry, airtight, and labeled containers.
Step 8: Storage
The prepared formulations were stored at room temperature in a dry place away from moisture and direct sunlight.
Step 9: Further Evaluation
All formulation batches (F1, F2, and F3) were subjected to further physicochemical evaluation studies.
2.5 EVALUATION OF POLYHERBAL FACE PACK
The prepared formulations (F1, F2, and F3) were evaluated for various physicochemical and performance parameters.
2.5.1 Organoleptic Evaluation
Procedure:
Step 1: A small quantity of formulation was taken on a clean surface.
Step 2: Color and appearance were observed visually.
Step 3: Odor was determined by smelling the sample.
Step 4: Texture was evaluated by rubbing powder between fingers.
Parameters:
• Color
• Odor
• Texture
•Appearance
2.5.2 pH Determination
Procedure:
Step 1: 1 g formulation was mixed with 10 mL distilled water.
Step 2: The mixture was stirred for 5 minutes.
Step 3: pH was measured using digital pH meter.
2.5.3 Spreadability
Procedure:
Step 1: The formulation was mixed with water to prepare paste.
Step 2: The paste was placed between two glass slides.
Step 3: Weight was placed on upper slide.
Step 4: Time required for movement of slide was recorded.
Formula:
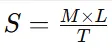
2.5.4 Washability
Procedure:
Step 1: The formulation paste was applied on skin surface.
Step 2: After drying, it was washed with water.
Step 3: Ease of washing was observed.
2.5.5 Skin Irritation Test
Procedure:
Step 1: Small quantity of formulation was applied on forearm.
Step 2: The formulation was kept for 15–20 minutes.
Step 3: Skin was observed for redness, itching, or irritation.
2.5.6 Drying Time
Procedure:
Step 1: The formulation was mixed with water to form smooth paste.
Step 2: The paste was applied as thin layer on skin.
Step 3: Time required for complete drying was recorded using stopwatch.
2.5.7 Flow Properties
(a) Angle of Repose
Procedure:
Step 1: About 10 g powder was allowed to flow through funnel.
Step 2: Height and radius of heap were measured.
Formula:
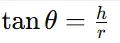
(b) Carr’s Index
Procedure:
Step 1: Bulk density and tapped density values were recorded.
Step 2: Carr’s index was calculated using standard formula.
Formula:

(c) Hausner Ratio
Procedure:
Step 1: Bulk density and tapped density values were used.
Step 2: Hausner ratio was calculated.
Formula:
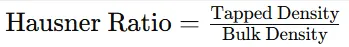
2.5.8 Stability Study
Procedure:
Step 1: Formulations were packed in airtight containers.
Step 2: Samples were stored at room temperature and accelerated conditions.
Step 3: Samples were evaluated at regular intervals.
Step 4: Color, odor, texture, appearance, and pH were observed and recorded.
3. RESULTS AND DISCUSSION
3.1. RESULTS OF PRE-FORMULATION STUDIES
3.1.1. Organoleptic Evaluation
Table 4: Organoleptic Evaluation of Herbal Ingredients
|
Ingredient |
Color |
Odor |
Texture |
|
Multani Mitti |
Light brown |
Characteristic earthy odor |
Smooth powder |
|
Neem Powder |
Greenish brown |
Characteristic neem odor |
Fine powder |
|
Orange Peel Powder |
Orange brown |
Citrus odor |
Slightly coarse |
|
Linseed Powder |
Brown |
Mild nutty odor |
Fine powder |
|
Activated Charcoal |
Black |
Odorless |
Very fine powder |
|
Licorice Powder |
Yellowish brown |
Mild sweet odor |
Fine powder |
|
Green Tea Powder |
Dark green |
Characteristic tea odor |
Fine powder |
|
Aloe Vera Powder |
Light green |
Mild herbal odor |
Fine powder |
|
Tulsi Powder |
Green |
Characteristic odor |
Fine powder |
|
Manjistha Powder |
Reddish brown |
Mild earthy odor |
Fine powder |
|
Sandalwood Powder |
Light brown |
Pleasant odor |
Fine powder |
3.1.2. Particle Size Analysis
Table 5: Particle Size Analysis of Herbal Powder
|
Sieve Number |
Amount Retained (%) |
|
20 mesh |
8% |
|
40 mesh |
22% |
|
60 mesh |
60% |
|
Passing |
10% |
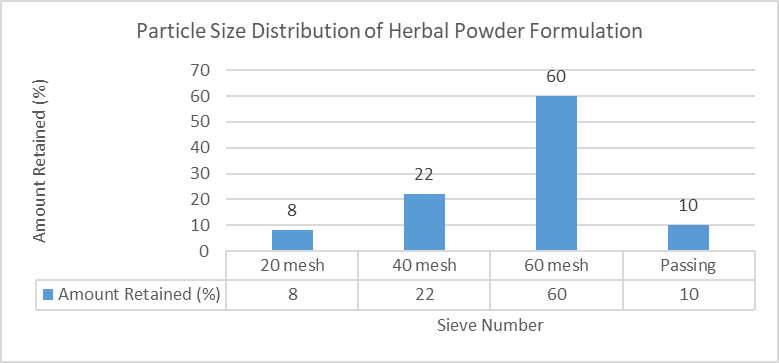
Figure 1: Particle size distribution of herbal powder formulation based on sieve analysis
Majority of powder passed through 60 mesh sieve, indicating uniform particle size.
3.1.3. Bulk Density
Table 6: Bulk Density of Powder Blend
|
Parameter |
Result |
|
Weight of powder |
5 g |
|
Bulk volume |
8 ml |
|
Bulk density |
0.62 g/ml |
3.1.4. Tapped Density
Table 7: Tapped Density of Powder Blend
|
Parameter |
Result |
|
Weight of powder |
5 g |
|
Tapped volume |
7 ml |
|
Tapped density |
0.71 g/ml |
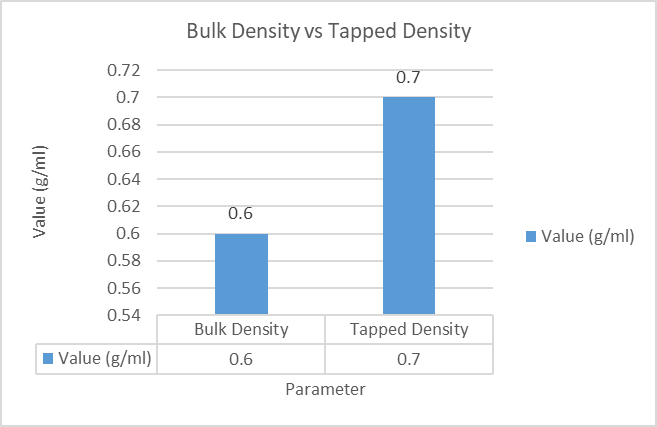
Figure 2: Comparative Graph of Bulk Density and Tapped Density of Polyherbal Face Pack Powder
3.1.5. Angle of Repose
Table 8 : Angle of Repose of Powder Blend
|
Parameter |
Result |
|
Height of powder cone |
3.5 cm |
|
Radius |
6.0 cm |
|
Angle of repose |
30.2° |
Interpretation: Powder shows good flow property.
3.1.6. Carr’s Index
Table 9: Carr’s Index of Powder Blend
|
Parameter |
Result |
|
Bulk Density |
0.62 g/ml |
|
Tapped Density |
0.71 g/ml |
|
Carr’s Index |
12.67 % |
Interpretation: Indicates good flowability.
3.1.7. Hausner Ratio
Table 10 : Hausner Ratio of Powder Blend
|
Parameter |
Result |
|
Tapped Density |
0.71 g/ml |
|
Bulk Density |
0.62 g/ml |
|
Hausner Ratio |
1.14 |
Interpretation: Indicates good powder flow property.
3.1.8. Moisture Content
Table 11: Moisture Content of Powder Blend
|
Parameter |
Result |
|
Initial weight |
2 g |
|
Final weight |
1.92 g |
|
Moisture content |
4 % |
Interpretation: Moisture content within acceptable limit.
3.1.9. pH Determination
Table 12: pH Determination of Formulation
|
Parameter |
Result |
|
Sample weight |
1 g |
|
Distilled water |
10 ml |
|
Observed pH |
6.2 |
Interpretation: Suitable for skin application.
3.1.10. Compatibility Study
Table 13: Compatibility Study of Herbal Ingredients
|
Observation Parameter |
Result |
|
Color change |
Not observed |
|
Odor change |
Not observed |
|
Lump formation |
Not observed |
Conclusion: No incompatibility observed between herbal ingredients.
3.1.11. Phytochemical Analysis
Table 14: Phytochemical Analysis of Herbal Ingredients
(+) → Present (–) → Absent
|
Sr. No. |
Phytochemical Constituents |
Observation |
Result |
|
1 |
Alkaloids |
Cream precipitate formed |
+ |
|
2 |
Flavonoids |
Pink / red color developed |
+ |
|
3 |
Tannins |
Blue-black / green color |
+ |
|
4 |
Saponins |
Persistent foam observed |
+ |
|
5 |
Glycosides |
Brown ring formed |
+ |
|
6 |
Phenolic compounds |
Dark green color |
+ |
3.2 RESULTS OF EVALUATION PARAMETERS
3.2.1 Organoleptic Properties of Formulation
Table 15: Organoleptic Evaluation of Face Pack
|
Batch |
Color |
Odor |
Texture |
Appearance |
|
F1 |
Light brown |
Herbal odor |
Smooth |
Fine powder |
|
F2 |
Brown |
Pleasant odor |
Smooth |
Uniform powder |
|
F3 |
Dark brown |
Mild odor |
Slightly soft |
Fine powder |
All formulations showed acceptable organoleptic properties. F2 showed better uniformity and pleasant odor compared to F1 and F3.
3.2.2 pH of Formulation
Table 16 : pH of Face Pack
|
Batch |
pH |
|
F1 |
6.0 |
|
F2 |
6.2 |
|
F3 |
6.5 |
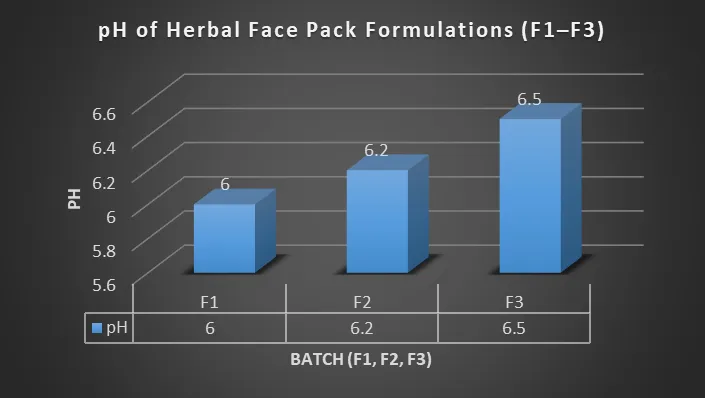
Figure 3: pH evaluation of herbal face pack formulations
All formulations were within the skin-compatible range (5–7). F2 showed ideal pH, making it most suitable for topical application.
3.2.3 Spreadability
Table 17 : Spreadability
|
Batch |
Spreadability (g·cm/sec) |
|
F1 |
18 |
|
F2 |
22 |
|
F3 |
20 |
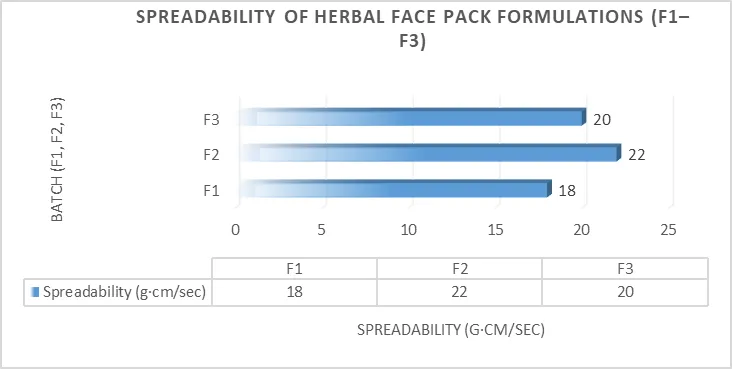
Figure 4 : Spreadability evaluation of herbal face pack formulations
F2 showed maximum spreadability indicating ease of application and better consistency.
3.2.4 Washability
Table 18 : Washability
|
Batch |
Observation |
|
F1 |
Easy |
|
F2 |
Easy |
|
F3 |
Moderate |
F1 and F2 were easily washable, whereas F3 showed slightly difficult removal due to higher moisture content.
3.2.5 Skin Irritation Test
Table 19 : Irritation Study
|
Batch |
Observation |
|
F1 |
No irritation |
|
F2 |
No irritation |
|
F3 |
No irritation |
All formulations were found to be non-irritant and safe for skin application.
3.2.6 Drying Time
Table 20 : Drying Time
|
Batch |
Time (min) |
|
F1 |
12 min |
|
F2 |
15 min |
|
F3 |
18 min |
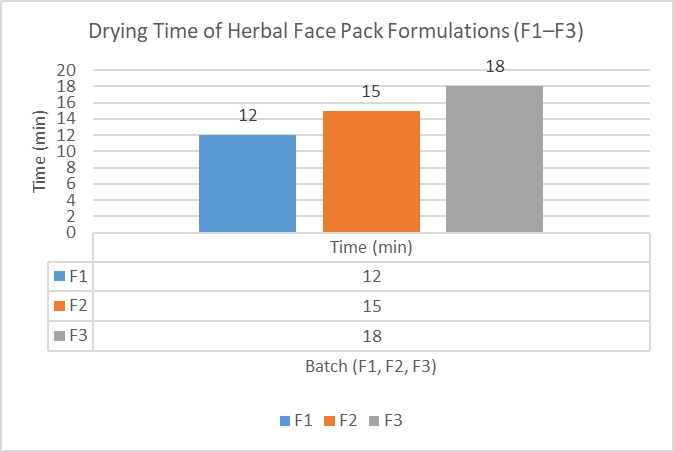
Figure 4: Drying time evaluation of herbal face pack formulations
F1 showed faster drying due to higher clay content, while F3 took longer due to higher moisturizing agents.
3.2.7 Stability Study
Table 21: Stability Study
|
Sr. No. |
Parameter |
Initial (0 Day) |
7 Days |
15 Days |
30 Days |
|
1 |
Color |
No change |
No change |
No change |
No change |
|
2 |
Odor |
No change |
No change |
No change |
No change |
|
3 |
pH |
6.5 |
6.5 |
6.4 |
6.5 |
|
4 |
Physical appearance |
Smooth |
Smooth |
Smooth |
Smooth |
3.3 DISCUSSION
The present study was carried out to formulate and evaluate a polyherbal face pack using different herbal ingredients for anti-acne and skin brightening effects. Preformulation studies showed that all selected herbal powders possessed acceptable physicochemical properties including good flow behavior, suitable particle size, acceptable moisture content, and skin-compatible pH, indicating their suitability for formulation development. Compatibility studies also confirmed that all ingredients were compatible with each other without any significant interaction.
The prepared formulations (F1, F2, and F3) exhibited acceptable physicochemical characteristics. Among all batches, formulation F2 showed better performance with optimum spreadability, good consistency, acceptable washability, and skin-friendly pH. The spreadability study indicated easy application of the formulation on the skin surface, while the washability study confirmed convenient removal after use. All formulations were found to be non-irritant during skin irritation studies, indicating safety for topical application.
The stability study demonstrated that the formulations remained stable under different storage conditions without significant changes in color, odor, texture, or pH. The improved performance of the formulation may be attributed to the synergistic action of herbal ingredients. Multani mitti helped in oil absorption and cleansing, neem and tulsi provided antimicrobial activity, while aloe vera, sandalwood, orange peel, and licorice contributed to moisturizing, soothing, antioxidant, and skin brightening effects. Overall, the study confirmed that the developed polyherbal face pack possesses good stability, safety, anti-acne activity, and skin-enhancing properties suitable for cosmetic application.
4. CONCLUSION
The present study was successfully carried out to formulate and evaluate a polyherbal face pack for anti-acne and skin brightening effects using natural herbal ingredients. The selected herbal ingredients such as multani mitti, neem, orange peel, aloe vera, tulsi, sandalwood, licorice, green tea, and activated charcoal were chosen due to their antimicrobial, anti-inflammatory, antioxidant, cleansing, and skin brightening properties. Preformulation studies confirmed that all herbal powders possessed suitable physicochemical properties including good flowability, acceptable particle size, compatibility, and skin-friendly pH. The formulations (F1, F2, and F3) were successfully prepared by dry mixing method and evaluated for various parameters such as pH, spreadability, washability, drying time, skin irritation, and stability.
Among all formulations, F2 showed the best performance with optimum spreadability, good consistency, easy washability, ideal pH, and better stability. The formulation was found to be non-irritant and suitable for topical application. The synergistic action of herbal ingredients contributed to effective cleansing, oil control, anti-acne activity, moisturizing effect, and improvement in skin brightness. Overall, the developed polyherbal face pack was found to be safe, stable, effective, and economical for skin care application. The study highlights the potential of herbal cosmetics as a natural alternative to synthetic cosmetic products with minimal side effects and good consumer acceptability.
REFERENCES


Shubhi Shukla, Anand Rai, Development Of Polyherbal Face Pack For Anti-Acne And Skin Brightening Effects, J. Pharm. Sci., 2026, 2 (5), 413-426. https://doi.org/10.5281/zenodo.20175258
 10.5281/zenodo.20175258
10.5281/zenodo.20175258